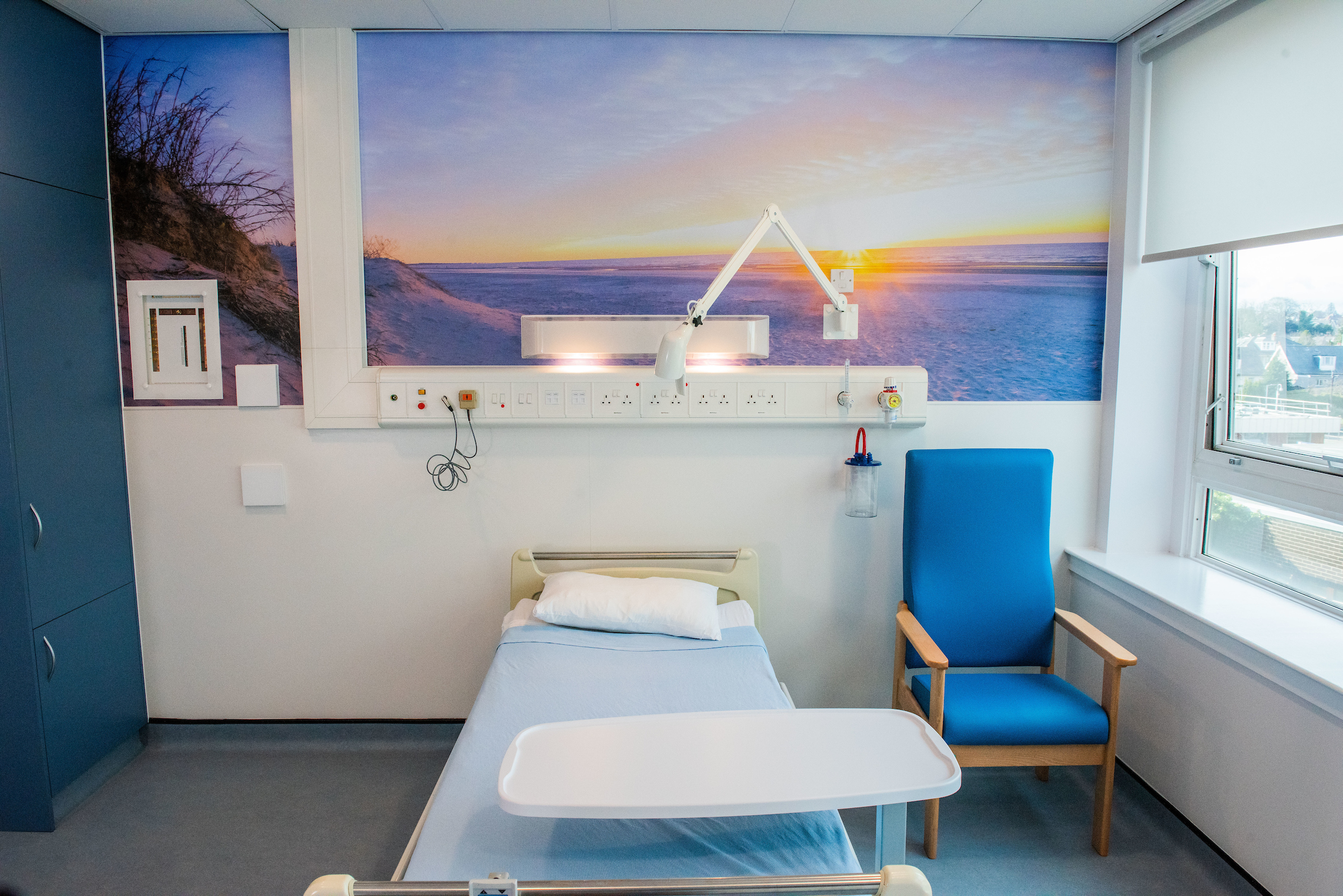
Edinburgh are delighted with their RAI Suite!
If your room needs an update please contact us.





For Help & Support Call 07399564463
or email amy@butterfly.org.uk
Get in touch with our patient support contacts
For all fundraising enquiries, or to order DVD’s, leaflets, TSH suppression cards and tee shirts please email us: enquiries@butterfly.org.uk
Edinburgh are delighted with their RAI Suite!
If your room needs an update please contact us.






Latest advice for patients having RAI Treatment Updated September 2020
Now that we are a few months in to the COVID-19 pandemic and have a greater understanding of the disease and it’s trajectory we have updated our guidance on RAI treatment. It is anticipated that the pandemic will result in services being disrupted for many months to come.
We have written the following guidance taking in to account both the risk patients face from cancer and from COVID-19 infection.
We recommend that radioactive iodine treatments can continue provided the local prevalence of COVID-19 is low and there is the appropriate infrastructure, staffing and COVID-19 testing available.
We appreciate there will be challenges in delivering a RAI service uniformly across the UK due to the differences in COVID-19 prevalence and its impact on local cancer centres so there will be inevitable variation in RAI services.
To reduce the risk of COVID-19 infection manifesting in the peri-RAI period:
Low iodine diet
Inpatient stay
Day case treatment
Subject to radiation protection risk assessment it is possible to deliver 1.1GBq I131 on a day case basis. This has the advantage of avoiding an inpatient stay and where feasible may also allow increased capacity whilst a backlog of patients is treated. It is recommended that similar precautions are taken to minimise the risk of COVID19 infection around the time of treatment.
Low-risk patients: (adjuvant setting)
High-risk patients (metastatic disease)
The recommendations have taken in to account discussions amongst UK thyroid cancer clinicians as well as general COVID-19 oncological advice at local and national levels.
Professor Jon Wadsley
Dr Laura Moss
Thyroid Awareness Month
Thyroid cancer is on the rise. It’s a non-smoking related condition that can affect people of any age. Lorna Nickson Brown discovered she had thyroid cancer after she found a lump in her neck. Don’t put your neck on the line. If you find a lump, get it checked by a doctor.
Please take our survey on the impact of COVID 19 for Thyroid Cancer Patients.
Dear Colleagues:
Thyroid Cancer: Radioactive Iodine Treatment during COVID-19 pandemic (2)
Now that we are a few weeks in to the COVID-19 pandemic and have a greater understanding of the disease and it’s trajectory we have revised our guidance on RAI treatment. It is anticipated that the pandemic will result in services being disrupted for many months to come so we cannot defer RAI treatment until the pandemic has completely passed.
We have written the following guidance taking in to account both the risk patients face from cancer and from COVID-19 infection.
We recommend that radioactive iodine treatments can be restarted provided the local prevalence of COVID-19 is low and there is the appropriate infrastructure, staffing and COVID-19 testing available. We appreciate there will be challenges in restarting a RAI service uniformly across the UK due to the differences in COVID-19 prevalence and its impact on local cancer centres so there will be variation in the resumption of RAI services.
To reduce the risk of COVID-19 infection manifesting in the peri-RAI period:
Low Iodine Diet
rhTSH (recombinant human TSH, Thyrogen) Preparation
Inpatient Stay
Day case treatment
Low risk patients: (adjuvant setting)
High risk patients (metastatic disease):
The recommendations have taken in to account discussions amongst UK thyroid cancer clinicians as well as general COVID-19 oncological advice at local and national levels.
Professor Jon Wadsley
Dr Laura Moss
We are aware that many patients with cancer have already received generic advice from the Chief Medical Officer regarding ‘shielding’ during the COVID-19 pandemic. The advice given below has been developed by UK thyroid cancer doctors and is tailored to specific thyroid cancer scenarios.
(i) Patients who have completed treatment for thyroid cancer
Patients who have previously received treatment for thyroid cancer such as surgery, with or without radioiodine (remnant ablation or radioiodine therapy), are not considered at higher risk of infection from COVID-19.
(ii) Patients in whom surgery for thyroid cancer is planned
Patients with significant symptoms and/or rapidly progressive disease should still be able to access surgery. For patients with cancers categorised as ‘low risk’ (where progression is considered to be slow) surgery may be delayed until a time when it is considered safer to proceed. Surgery may also be deferred for patients considered susceptible to severe COVID-19 infection or who may need intensive care monitoring after surgery. There will be variability in the provision of surgery across the UK due to the number of local COVID-19 patients and staff availability
(iii) Radioactive iodine therapy
Many centres ceased radioactive iodine treatment in the early days of the pandemic due to a combination of increased admissions and COVID-19 safety concerns.. In most cases, radioiodine therapy is not urgent and can be safely delayed. However, now that the acute pressures on the service are easing, centres are beginning to plan to restart this treatment. How soon it is possible to do this safely will depend on local circumstances. It is possible that patients will be advised to self-isolate and screened for COVID-19 prior to treatment to minimise the risk of becoming unwell following treatment. If you have concerns please discuss with your own hospital team.
(iv) TSH suppressive therapy
Patients on suppressive doses of thyroxine (i.e have a TSH target of <0.1mU/l) should continue on their current dose. Being on suppressive dose of levothyroxine does not increase the risk of COVID-19 infection.
(v) Multikinase inhibitors and chemotherapy
Patients who are receiving multikinase inhibitors (such as Lenvatinib or Sorafenib), or chemotherapy are at increased risk of severe illness from coronavirus and should follow government advice regarding shielding- https://www.gov.uk/government/publications/guidance-on-shielding-and-protecting-extremely-vulnerable-persons-from-covid-19/guidance-on-shielding-and-protecting-extremely-vulnerable-persons-from-covid-19.
They should expect to hear from their centre and to have a discussion about the advisability of continuing treatment at this time in their particular circunmstances.
(vi) Previous radiotherapy
Patients who have previously received external beam radiotherapy to the neck may be at increased risk of severe illness with coronavirus and should also consider self-isolating.
V3.0 2nd May 2020
COVID-19 Advice for patients with thyroid cancer
(i) Patients who have completed treatment for thyroid cancer
Patients who have previously received treatment for thyroid cancer such as surgery, with or without radioiodine (remnant ablation or radioiodine therapy), are not considered at higher risk of infection from COVID-19.
(ii) Patients in whom surgery for thyroid cancer is planned
NHS England has stated that surgery for cancer should continue at present, although elective surgery is cancelled in many hospitals. More detailed advice about which operations should be prioritised will shortly be released to surgeons across the UK.
(iii) Radioactive iodine therapy
Patients awaiting radioiodine therapy may well find that their treatment is postponed. Whilst the treatment itself does not increase the risk of infection, subsequent radiation protection issues would seriously complicate the care of any patient who subsequently became unwell with COVID-19. In most cases radioiodine therapy is not urgent and can be safely delayed. If you have concerns please discuss with your own hospital team.
(iv) TSH suppressive therapy
Patients on suppressive doses of thyroxine (i.e have a TSH target of <0.1mU/l) should continue on their current dose. Being on suppressive dose of levothyroxine does not increase the risk of COVID-19 infection.
(v) Multikinase inhibitors and chemotherapy
Patients who are receiving multikinase inhibitors (such as Lenvatinib or Sorafenib), or chemotherapy are at increased risk of severe illness from coronavirus and should follow government advice regarding shielding- https://www.gov.uk/government/publications/guidance-on-shielding-and-protecting-extremely-vulnerable-persons-from-covid-19/guidance-on-shielding-and-protecting-extremely-vulnerable-persons-from-covid-19.
They should expect to hear from their centre and to have a discussion about the advisability of continuing treatment at this time in their particular circunmstances.
(vi) Previous radiotherapy
Patients who have previously received external beam radiotherapy to the neck may be at increased risk of severe illness with coronavirus and should also consider self-isolating.
v1.0 23rd March 2020
To be reviewed in 2 weeks
Please leave a message on our answer phone and we will get back to you as soon as we can.
Email: amy@butterfly.org.uk
Scan our QR code to donate today!

 Excellence in Oncology
Excellence in Oncology 'Bright Ideas'
'Bright Ideas' Excellence in Oncology
Excellence in Oncology Highly Recommended
Highly Recommended